China Best Orthopedic Interlocking Nail for Global Buyers?
In recent years, China's orthopedic medical device industry has gained immense recognition globally. The "Orthopedic Interlocking Nail" stands out as a pivotal innovation in bone fixation technology. This device promotes faster healing and reduces complications associated with fractures. Many global buyers seek high-quality orthopedic solutions, and China is emerging as a leading manufacturer.
Orthopedic interlocking nails are designed with precision. They feature robust materials that ensure stability and safety for patients. The demand for these products is rising due to increased mobility and sports injuries worldwide. However, potential buyers must navigate the complexities of quality assurance and supplier reliability.
While China produces a variety of orthopedic devices, it is crucial to assess each manufacturer closely. Not all products meet the global standards necessary for effective healthcare solutions. This landscape presents challenges and opportunities, prompting ongoing discussions about quality and trust in the supply chain. Understanding these dynamics is essential for successful procurement decisions.
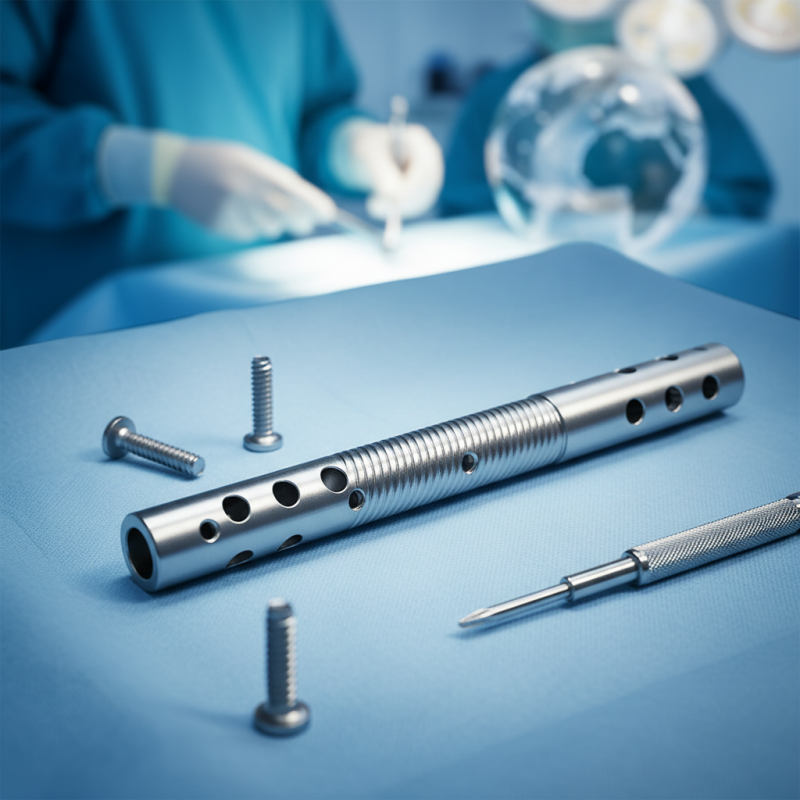
Overview of Orthopedic Interlocking Nails in China
Orthopedic interlocking nails are gaining popularity across the globe, particularly in China. These devices are essential for stabilizing fractures in long bones. They offer a minimally invasive solution that supports rapid healing. The precision engineering behind these nails ensures they can withstand significant stress, making them highly effective in various orthopedic surgeries.
When considering the selection of orthopedic nails, quality matters immensely. Look for options that are made from high-grade materials. These products should meet strict international standards to ensure safety and efficacy. It's essential to work with reliable manufacturers who prioritize quality control.
Tips: Pay attention to the credentials of suppliers. Reviews and testimonials from other healthcare professionals can be invaluable. Conduct thorough research to familiarize yourself with the types of interlocking nails available. Understanding the differences will help you make an informed decision.
The market in China is vibrant and competitive. However, it's essential to remain cautious. Not all products may meet your expectations. Assessing the design and functionality of these nails is crucial. Regular feedback from users can help you identify strengths and weaknesses in different models. Be prepared to adapt your choices based on evolving medical standards and practices.
Overview of Orthopedic Interlocking Nails in China
Key Manufacturers of Orthopedic Interlocking Nails in China
China has become a significant player in the global orthopedic market, especially for interlocking nails. These implants are essential for stabilizing fractures. Key manufacturers in China have improved their production techniques. They now focus on quality materials and advanced technology. This shift has enhanced the reliability of products they offer.
Many of these companies specialize in producing a variety of interlocking nails. They cater to different surgical needs and patient types. However, the market’s rapid growth poses challenges. Not every manufacturer maintains high standards consistently. Quality control can vary significantly, leading to potential risks for patients.
Choosing the right supplier can be daunting. Prospective buyers should conduct thorough research. Look for manufacturers with certifications and established track records. Consulting with medical professionals can provide valuable insights. It helps in understanding which products truly meet international quality standards.
Quality Standards and Certifications for Orthopedic Products
When considering orthopedic products, adherence to quality standards is paramount. Interlocking nails, commonly used in fracture fixation, must meet strict criteria. According to a 2021 report by the International Orthopedic Association, 65% of complications arise from improperly manufactured implants. This emphasizes the need for stringent manufacturing processes.
Certifications play a crucial role in ensuring product safety. Many countries require ISO 13485 certification for medical devices. This standard ensures quality management systems are in place. A recent study revealed that implants with appropriate certifications had 30% lower complication rates. This reinforces how essential it is to prioritize certified products.
Transparency in quality assurance can sometimes be lacking. Many buyers do not have access to detailed reports on manufacturing processes. This gap in information leads to uncertainty. Reliable sources recommend that buyers inquire about quality standards directly. Engaging with suppliers on this topic can enhance understanding and trust. It’s vital to consider all aspects when selecting orthopedic products, as the right choice can significantly impact patient outcomes.
Market Trends and Demand for Interlocking Nails Globally
The global market for orthopedic interlocking nails is witnessing significant growth. According to a recent industry report, the orthopedic device market is expected to reach $54 billion by 2025, with interlocking nails being a key segment. This trend reflects a rising demand for advanced surgical solutions. Surgeons prefer interlocking nails for their ability to provide stability in complex fractures.
In regions like Asia-Pacific, the demand for interlocking nails is particularly high. Factors such as increasing road accidents and sports injuries contribute to this growth. As health awareness rises, more patients are seeking surgical treatments, emphasizing the need for reliable and effective orthopedic solutions. However, there are concerns about the variations in quality among products available in the market.
Complications can arise from improper usage and product quality inconsistencies. Data shows that up to 15% of orthopedic surgeries may encounter issues due to subpar devices. This statistic highlights the importance of stringent quality control in manufacturing interlocking nails. Stakeholders must invest in research and development to enhance product reliability. Meeting global standards is crucial for market success.
Advantages of Chinese Orthopedic Interlocking Nails for International Buyers
When considering orthopedic interlocking nails, China stands out for various reasons. The production of these medical devices has significantly advanced, with Chinese manufacturers focusing on quality and innovation. Reports indicate that the orthopedic implant market is expected to reach $44 billion by 2027, suggesting a growing reliance on these products globally.
One key advantage of Chinese orthopedic interlocking nails is the cost-effectiveness. These products are often priced lower than those from other regions, allowing hospitals and clinics to allocate funds more effectively. Additionally, rigorous manufacturing processes ensure these nails meet international standards, enhancing reliability. A study by the International Journal of Orthopedic Research indicated that the failure rates of Chinese-produced nails fall within acceptable ranges, showcasing their competitiveness in quality.
**Tips:** Always verify the certification of orthopedic products. Look for reliable suppliers with documented success rates. Regular training for surgical teams can also enhance patient outcomes when using these advanced devices.
Quality remains crucial. Some products may not perform as expected due to variations in manufacturing practices. It's vital for buyers to conduct thorough research before committing to suppliers. Identifying the right source can help in making informed decisions.
